Purpose of Electrolytes
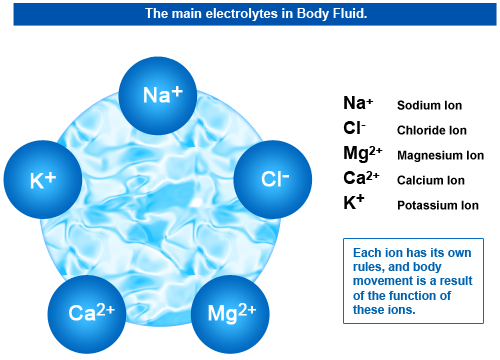
The Structure of ElectrolytesElectrolytes are minerals in your blood and other bodily fluids that carry an electric charge (hence the name). They consist of ions, which are atoms that have either a negative or positive charge. An electrolyte that is dilute has a relatively small amount of ions for its volume, while one that is concentrated has a large amount of ions ("What is an Electrolyte," n.d.). They can be acids, bases, and salts and are replaced by drinking fluids. Common electrolytes include sodium, potassium, calcium, magnesium, chloride, and phosphorous.
|
The Role of Electrolytes in the BodyElectrolytes are vital for humans to survive, helping to regulate the amount of water in the body, the acidity of the blood, how nerves and muscles function, blood pressure, and the rebuilding of damaged tissue. Our neurons and muscles are perceived as electric tissues of the body, meaning they are activated by the electrolyte activity in the fluids inside and outside of the cells. For example, a muscle contraction needs calcium, sodium, and potassium to be present. Our heart, nerve and muscle cells use electrolytes to maintain voltages across their cell membranes and to carry electrical impulses across themselves to other cells (Nordqvist, 2009). Electrolytes need to be replenished after physical exercise because you lose them through sweat.
|